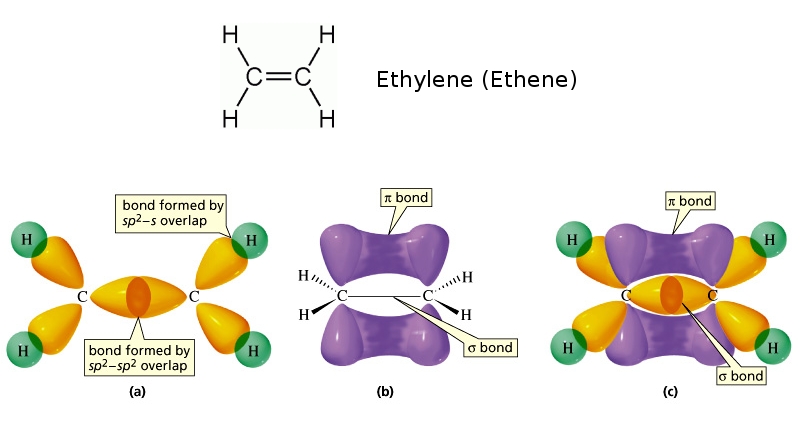
In an SP2 hybrid orbital than an SP3 hybrid orbital and since the electronĭensity in an S orbital is closer to the nucleus. Two out of three gives us 67% P character. One out of three, gives us 33% S character in our new hybrid SP2 orbital and then we have two P orbitals. That we're taking here and one of them is an S orbital. In terms of what percentage character, we have three orbitals Once again, when we draw the pictures, we're going to ignore We're gonna take these orbitals and hybridized them to form three SP2 hybrid orbitals and they have a bigger front lobe and a smaller back lobe here like that. We know that a P orbital is shaped like a dumbbell. Our new hybrid orbital, let's go ahead and get Notice that we left a P orbital untouched. This carbon right here is SP2 hybridized and same with this carbon. This is SP2 hybridization because we're using one S Orbital and two P orbitals to form This is an SP2 hybrid orbital and same with this one,Īn SP2 hybrid orbital. Orbitals has one electron and it's like that. We're gonna take one of the P's and then another one of the P's here. 
We're gonna promote the S orbital up and this time, we only In this case, we only have a carbon bonded to three atoms. Orbitals and combined them to make four SP3 hybrid orbitals. In the video on SP3 hybridization, we took all four of these We're gonna start with ourĮlectron configurations over here, the excited stage. A hydrogen, a hydrogen and a carbon and so we must need aĭifferent hybridization for each of the carbon's presence in the ethylene molecule. Approximately, 120 degree bond angles and this carbon that I've underlined here is bonded to only three atoms. And the bond angles areĬlose to 120 degrees. You could think aboutĪll this in a plane here. Actually, this entire molecule is planar. The geometry of theĪtoms around this carbon happens to be planar. The carbons in ethenes, let's say this carbon right here, we don't see the same geometry. Is bonded to four atoms, we have an SP3 hybridization with a tetrahedral geometryĪnd an ideal bonding over 109.5 degrees. This allows each #O# atoms to have 1 unhybridized p-orbital with which to form a pi bond.Voiceover: In an earlier video, we saw that when carbon How many sigma and pi bonds in co2, On the other hand, each #O# atom has three regions of electron density around it, which means it is #sp^2# hybridized. 
In carbon dioxide molecule, a carbon atom is joined by four covalent bonds to two oxygen atoms, which have two covalent bonds each. The type of bond between atoms in a molecule of CO2 is polar covalent bond. Hence, Option(B) Two sigma and two pi bonds, is the correct option, so A CO2 molecule contains Two sigma and two pi bonds. What are the sigma bonds and pi bonds of CO2? The central atom carbon is doubly bonded to the two oxygen atoms. THEREFORE, A TOTAL OF 1 SIGMA BOND AND 2 PI BONDS ARE PRESENT IN CO. The bonds are formed by the 4 valence electrons in carbon and 6 in oxygen. How many pi and sigma bonds does Co have? Double bonds have one sigma and one pi bonds. Does carbon dioxide have two π bonds and σ bonds?Ĭarbon dioxide has two sigma and two pi bonds. What are the sigma bonds and pi bonds of CO2?Ĭarbon dioxide has two sigma and two pi bonds.Does carbon dioxide have two π bonds and σ bonds?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
